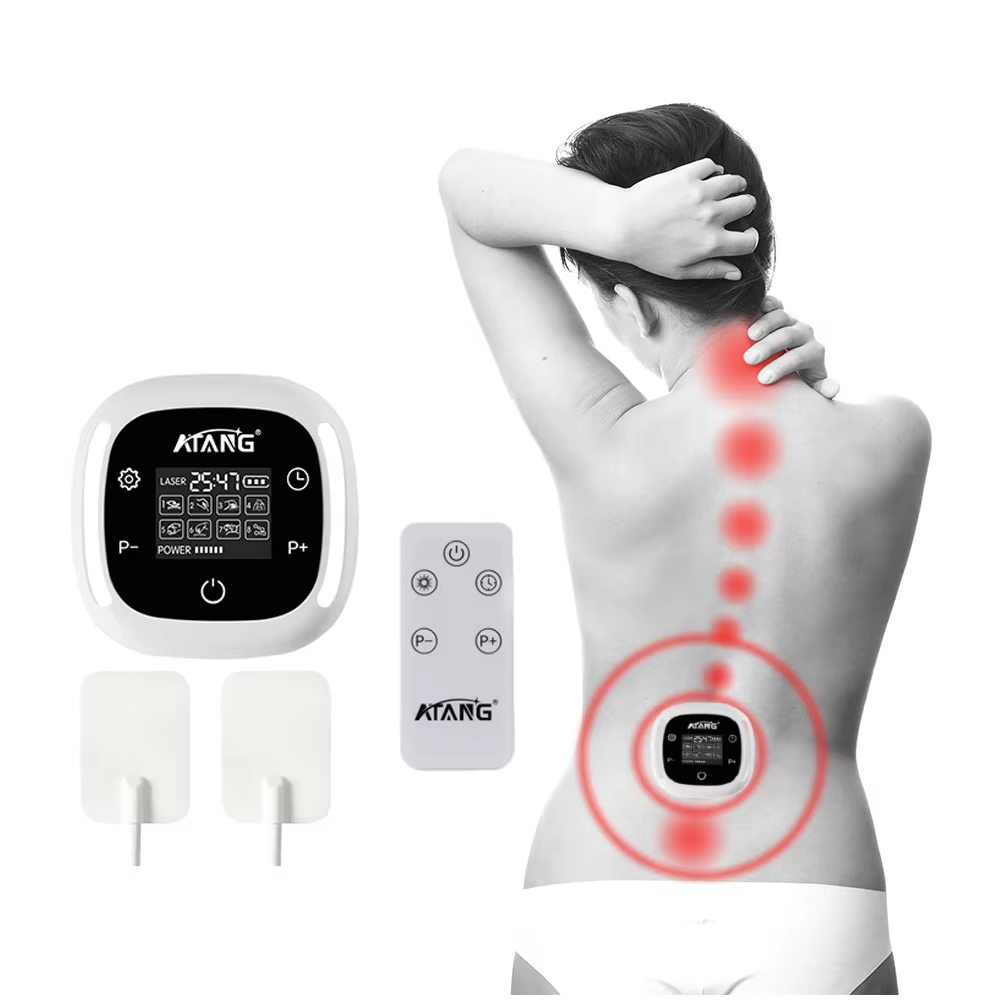
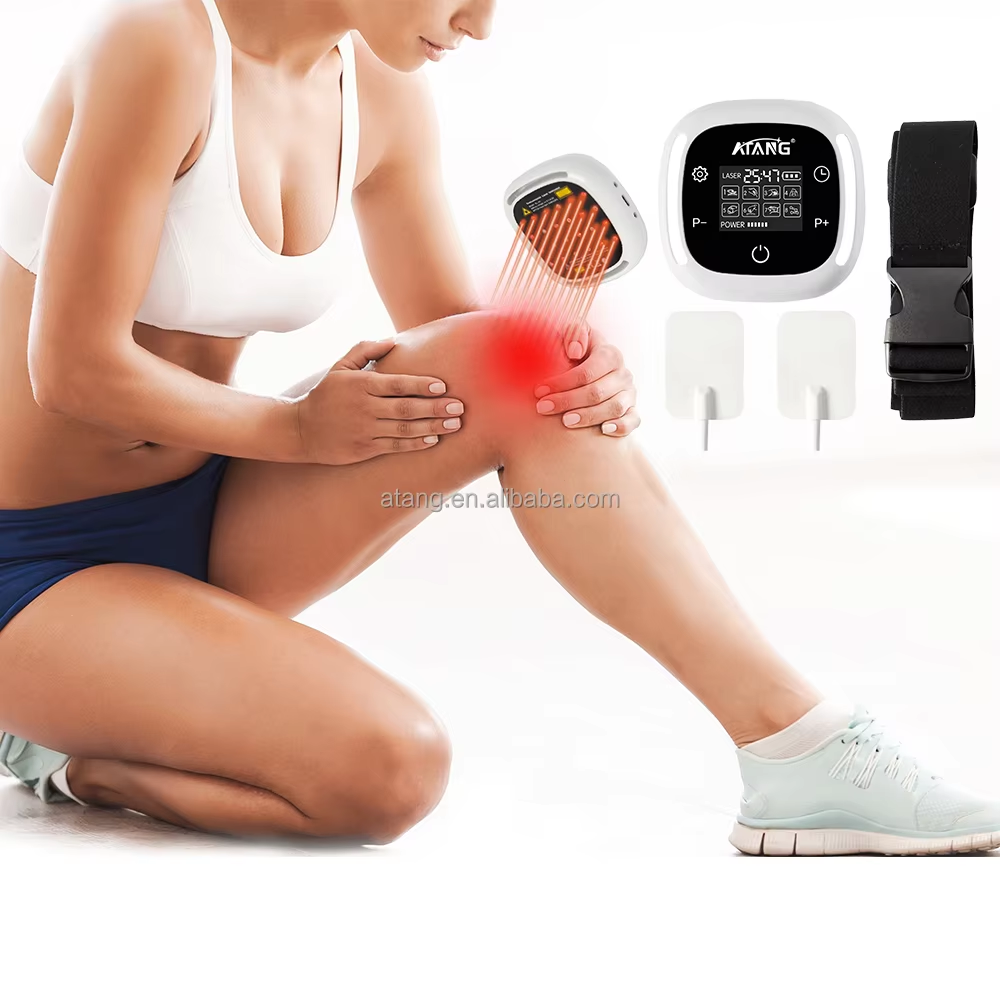
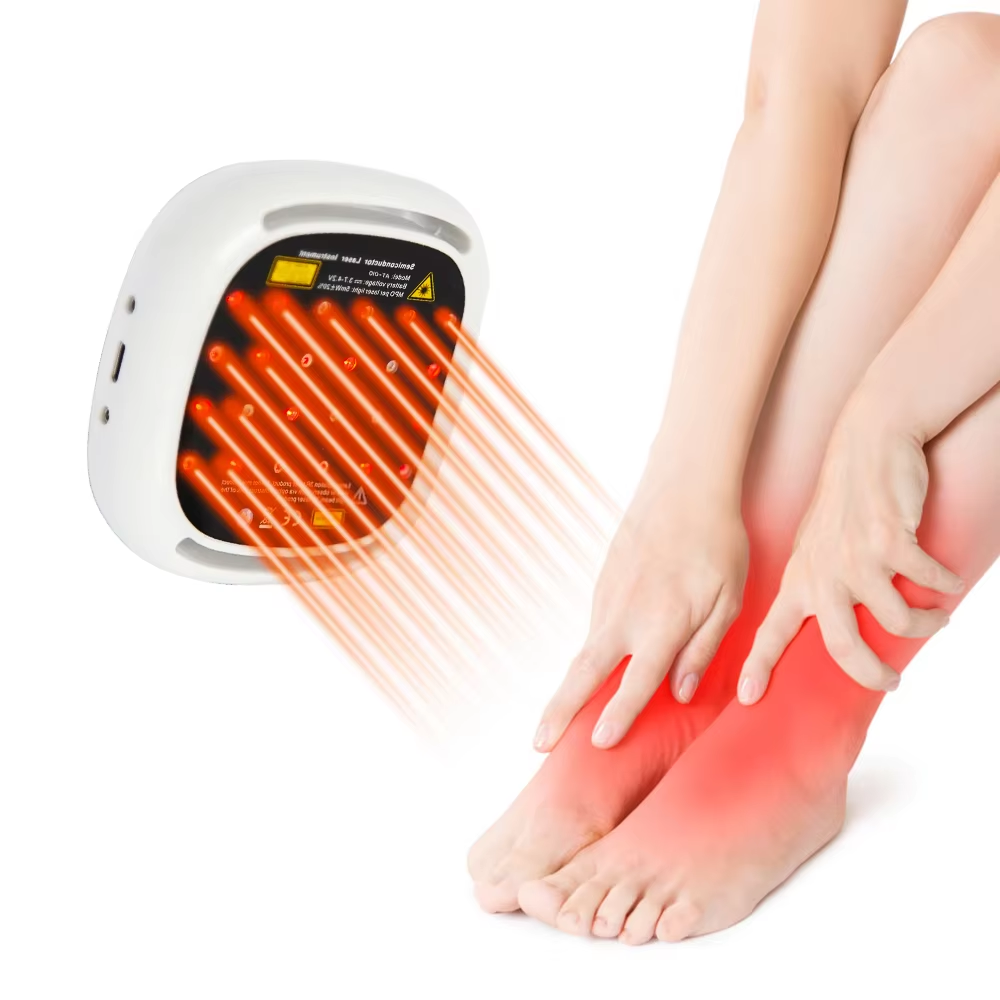
The pursuit of an effective and trustworthy solution for chronic lower back pain leads many to seek treatments that are not only innovative but also recognized by authoritative health regulatory bodies. For those prioritizing safety, quality, and a non-pharmacological approach, an FDA Cleared Lower Back Pain Treatment stands as a significant consideration. At ATANG, we bridge the gap between professional therapeutic technology and accessible home care through our Low-Level Laser Therapy (LLLT) device, developed within a regulatory framework that underscores our dedication to responsible healthcare innovation.
Understanding the designation "FDA Cleared" is crucial for informed decision-making. For medical devices, this typically means the product has been reviewed by the U.S. Food and Drug Administration and found to be substantially equivalent to a legally marketed predicate device. It is a regulatory step that involves demonstrating safety and effectiveness for its intended use. Our product is manufactured in a facility registered with the FDA (Registration #3015515517), which operates under a Quality System Regulation. This system governs the methods, facilities, and controls used in the design, manufacturing, packaging, labeling, storage, and servicing of devices. Therefore, when you choose this treatment, you are not just selecting a device; you are opting for a product developed within an ecosystem of documented procedures and quality controls, providing a foundational layer of assurance regarding its production standards and intended safety profile.
The core therapeutic mechanism of this treatment is Low-Level Laser Therapy, or photobiomodulation. This technology operates on a cellular level. The device delivers specific wavelengths of low-intensity laser light that penetrate the skin to reach the deeper tissues of the lower back. This light energy is primarily absorbed by mitochondria, the powerhouses within our cells. This absorption is believed to stimulate a series of biochemical events, leading to enhanced cellular metabolism and increased production of adenosine triphosphate (ATP), the fundamental energy currency of the cell. This boost in cellular energy can support various recovery processes: it may help reduce the production of pro-inflammatory cytokines, thereby alleviating inflammation; improve microcirculation to enhance the delivery of oxygen and nutrients while removing waste products; and modulate pain signals. For the individual suffering from lower back pain, this translates to a targeted, non-invasive intervention designed to address key underlying factors of discomfort—such as muscle strain, ligament issues, or joint inflammation—without generating heat or requiring any surgical intervention.
The practical application of this FDA Cleared treatment is designed for integration into daily life, offering a professional-grade tool for home use. Consider the individual with a sedentary profession, who experiences debilitating stiffness and ache by the end of the workday. This device provides a proactive way to manage that discomfort, with sessions that can be conveniently administered at home. For the active person or aging individual dealing with recurring discomfort from conditions like age-related joint changes, it serves as a maintenance tool to help manage symptoms and support mobility. The value lies in its dual role: as a potential acute intervention for flare-ups and as a part of a chronic management strategy, all within a framework of regulatory oversight that distinguishes it from general wellness products. It represents an empowerment tool, putting a level of clinical-grade technology into the user's hands.
Ultimately, our FDA Cleared Lower Back Pain Treatment embodies ATANG's mission to bring credible and healthy products to families worldwide. It merges the scientific principles of photobiomodulation with the rigor of regulatory compliance. We invite you to consider this advanced option for managing your lower back health. For detailed information on device specifications, clinical insights, and distribution opportunities, please contact our team directly to discuss how this treatment can meet your specific needs or business goals.