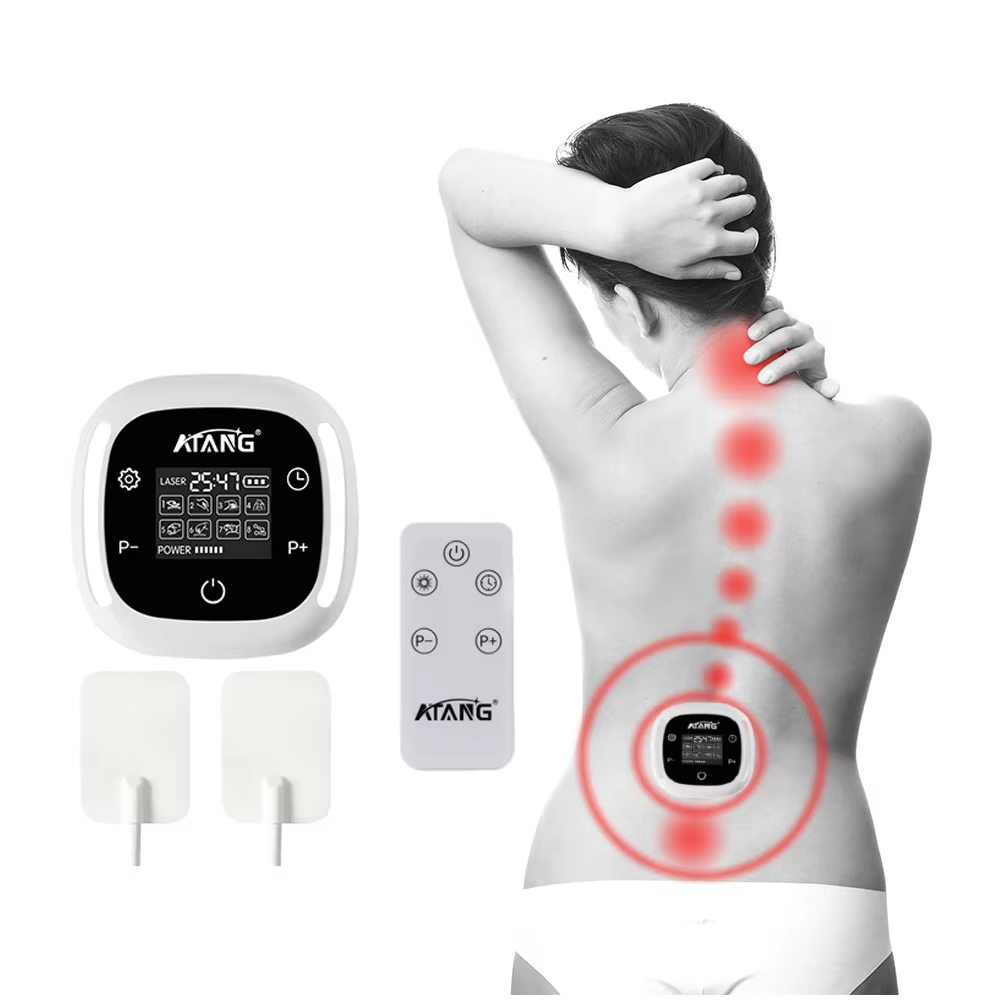
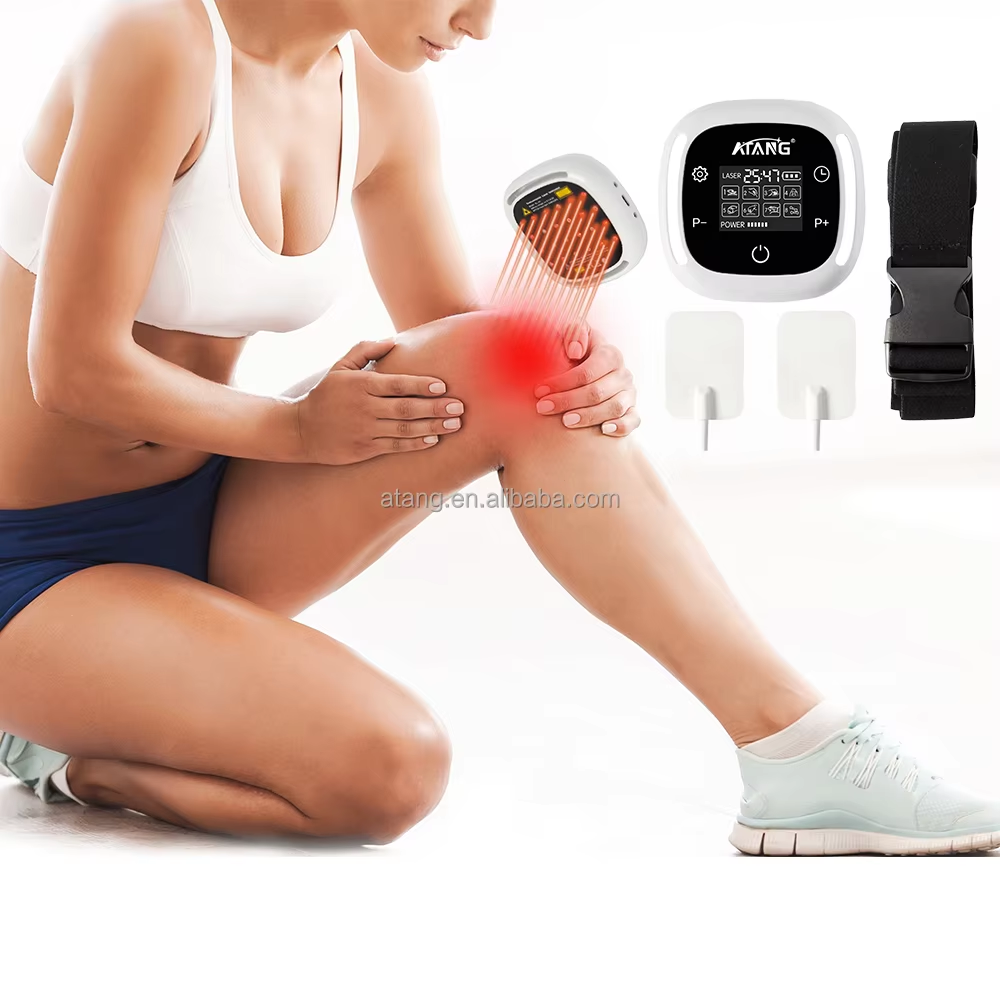
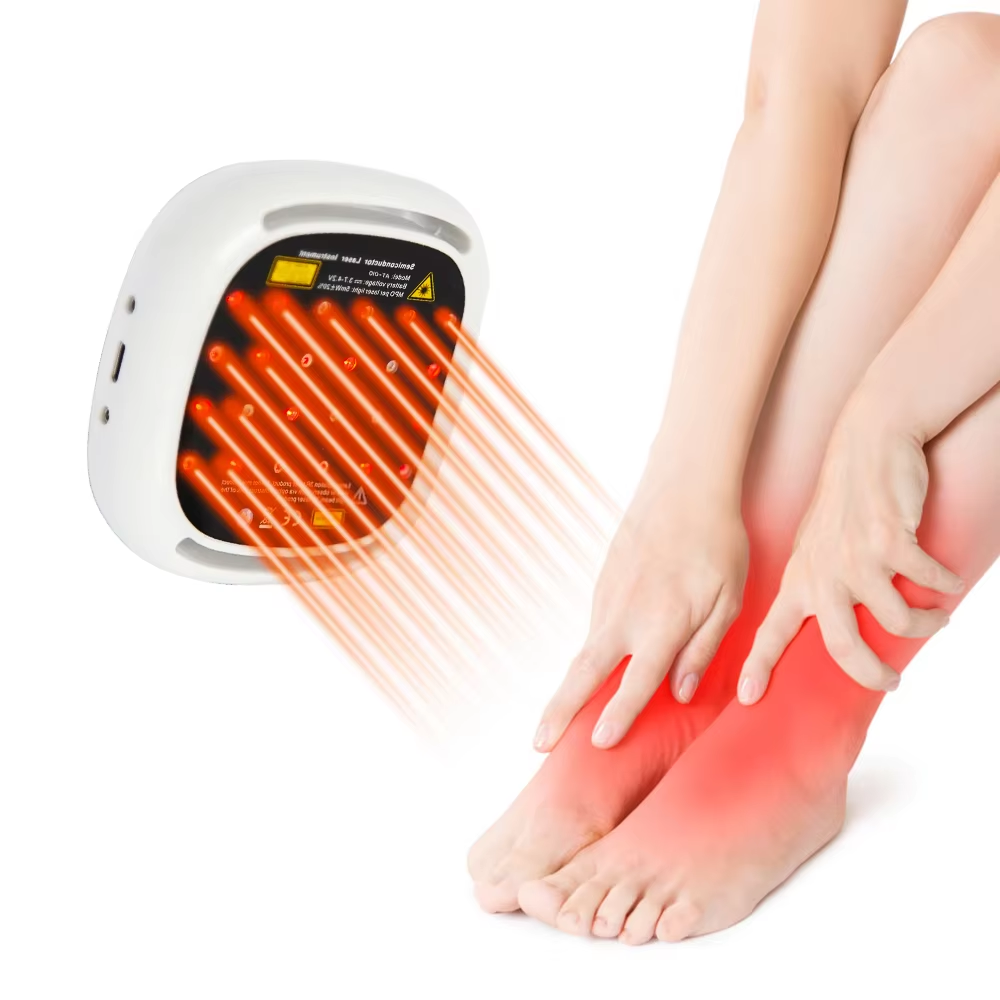
The search for a certified lower back pain device reflects a sophisticated and essential shift in consumer health awareness. Certification, particularly marks like CE (Conformité Européenne) and listings from bodies like the U.S. Food and Drug Administration (FDA), serve as critical gatekeepers in the medical device landscape. For a condition as prevalent and impactful as lower back pain—a leading cause of disability globally—these certifications are not mere administrative stickers. They are tangible assurances that a device has undergone a formal conformity assessment against strict safety, health, and environmental protection requirements. A CE-marked medical device, for example, complies with the European Union's Medical Devices Regulation, which governs everything from clinical evaluation and risk management to post-market surveillance. Similarly, an FDA-registered device comes from a facility that adheres to Current Good Manufacturing Practices. Therefore, a certified lower back pain device represents a product that has been legally deemed safe for its intended use and manufactured under controlled, auditable conditions. This is paramount, as it directly addresses consumer concerns about product safety, electrical integrity, and the accuracy of performance claims.
ATANG Health has embedded this philosophy of verified compliance into its core operations. Our claim as a provider of certified lower back pain devices is substantiated by our formal credentials. Our CE certification and FDA establishment registration (Number: 3015515517) are foundational pillars of our identity. They signal to distributors, healthcare professionals, and end-users that we operate within a structured, transparent framework of quality assurance. This is especially crucial for devices utilizing technologies like Low-Level Laser Therapy (LLLT). While LLLT is supported by a body of research indicating its benefits for musculoskeletal pain—with studies showing it can lead to statistically significant reductions in pain intensity for chronic low back pain sufferers—the safety of its application is dependent on proper engineering. Our independent SGS 60825 laser safety report provides an additional layer of verification, ensuring the optical output is within safe parameters for home use. This multi-layered approach to certification transforms our products from simple wellness gadgets into credible home-care instruments.
Consider the practical implications for an individual suffering from chronic, non-specific lower back pain. They are often navigating a maze of options, from over-the-counter medication to physical therapy. Introducing a home-use device into their management plan is a significant decision. Choosing a certified lower back pain device from ATANG mitigates key risks. The certification assures them that the device's materials are biocompatible, its electrical circuits are isolated and safe, and its laser output is non-hazardous. Beyond safety, certification implies reliability and consistency in performance—the device will deliver the same therapeutic dose each time it is used as intended. For a chronic condition requiring consistent management, this reliability is as important as initial efficacy. It empowers the user to build a routine with a tool they can trust, potentially reducing their reliance on more invasive or pharmaceutical interventions over time. The certification marks become a shorthand for this entire value proposition: safety, quality, and trustworthy performance.
It is important to contextualize the role of a certified lower back pain device. These products are designed as supportive aids for managing discomfort and promoting musculoskeletal wellness within a home setting. They are part of a comprehensive approach that should include professional medical advice, especially for acute or specific diagnoses. ATANG's role is to provide a high-quality, certified tool that fits into this broader picture. Our dedication to certification, combined with our investment in patented designs and clinical research, positions us as a serious manufacturer in the smart home healthcare sector. We aim to set a standard where advanced technology and rigorous safety compliance go hand in hand.
If you are a distributor, clinician, or individual seeking reliable and certified lower back pain devices for the market or personal use, we invite you to connect with us. Contact ATANG Health today to request detailed documentation on our certifications, device specifications, and clinical support materials. Let us provide you with the transparency and evidence needed to make a confident, informed decision.